The first gene therapy for LCA
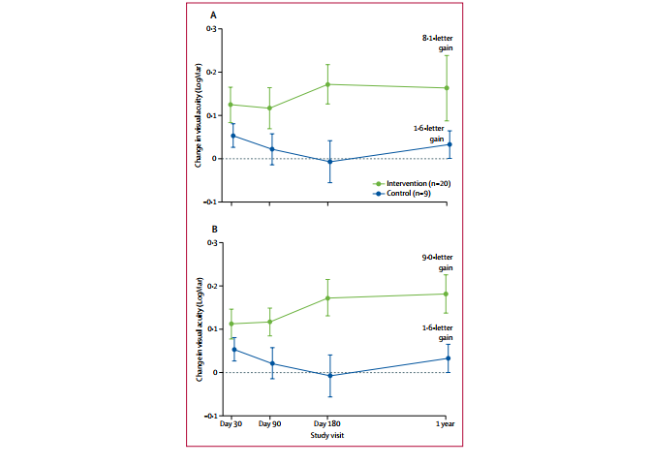
The 2017 Meeting of the American Academy of Ophthalmology (AAO) saw the presentation of the results of a phase III clinical trial run by Dr. Stephen R. Russell, the ophthalmologist of the University of Iowa. This involved the use of gene therapy in patients with Leber’s congenital amaurosis (LCA), a disease of a genetic origin that affects the retina. The condition manifests itself in the first years of life and progresses slowly until it causes total blindness. It affects 1 in every 80,000 people and has no current treatment approved by the FDA (US Food and Drug Administration).
The study shows how this gene therapy, the first of its type, can restore sight to people with this hereditary retinal disorder. The treatment called voratigene noparvovec (Luxturna, Spark Therapeutics) consists of an intraocular injection containing a genetically modified version of an inoffensive virus to carry the corrected version of the mutated gene to the patient’s retina.
93% of patients treated with Luxturna experienced significant improvements in their sight, enough to navigate a maze with a light of low-moderate intensity, which allows them to walk without a stick or a guide dog.
This gene therapy is currently being reviewed by the FDA for its possible approval in January 2018. This approval could pave the way for gene therapies in other retinal diseases such as retinitis pigmentosa.
Image: Russell, Stephen, et al. “Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial.” The Lancet 390.10097 (2017): 849-860.